The liver plays an important role in regulating energy homeostasis and drug metabolism. It produces thousands of substances and enzymes needed in the human body, including bile, and detoxifies a variety of toxic substances. The mechanism of drug-induced liver toxicity is closely related to liver metabolic activation. Research on the liver has been closely focused on by experts.
Although liver cells have almost unlimited regenerative capacity in the body, once removed from the body, liver cells only survive for two to three days and their unique replication function quickly disappears. With the rapid development of cell biology, it has been found that 3-D culture systems can significantly promote the maturation function of hepatocytes in vitro, suggesting that a 3-D culture environment provides the spatial location, pressure signal, and matrix adhesion information of cells, which plays an important role in the survival and function of hepatocytes.
Liver organoids can better simulate the metabolic and synthetic functions of the liver, such as glycogen metabolism, lipid metabolism, steroid metabolism, drug metabolism, albumin secretion, urea synthesis, etc. And they can highly express hepatocyte-related drug metabolism enzymes, nuclear receptors, and liver-specific markers, such as ALB, AAT, HNF-4α, HNF-3β, etc.
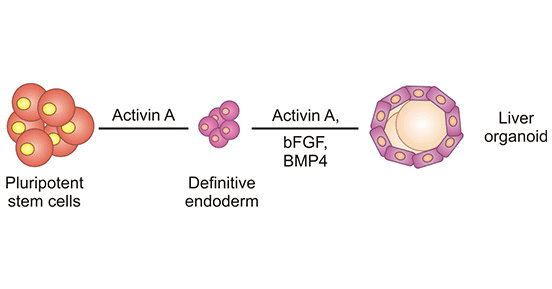
Figure 1. Establishment process of liver organoids
Liver organoids derived from healthy donors represent ideal models for understanding liver biological processes such as liver development and regeneration, modeling liver cancers and other liver diseases such as chronic and inherited diseases, as well as applying in regenerative medicine including drug response and transplantation. Gene editing, virus infection, and drug introduction to the liver organoids can simulate the occurrence and developmental processes of diseases. Normal liver organoids are ideal models for drug screening, translational research, as well as detoxification and metabolic studies in the clinic. Organoids coming from diseased samples, including cancer organoids, recapitulate the original characteristics of patients.
Table: Sources of liver organoids and their functions
| Sources of liver organoids | Function |
| iPSC | Albumin secretion, strong drug metabolism |
| MSC | Expression of early liver markers |
| iPSC, matrix cell | Liver-specific gene expression, albumin and urea secretion, cytochrome P450 activity |
| iPSC, SWISS, 3T3 cell | Hepatocyte related gene expression, albumin secretion, urea production |
| MSC, HUVEC | Liver specific gene expression and albumin secretion |
| Hepatocyte | Albumin secretion, high expression of liver-specific genes |
| Hepatoma carcinoma cell, MSC, HUVEC | High expression of liver-specific genes and urea formation |
| iPSC, MSC, HUVEC | High expression of albumin, tubular proteins, and other functional markers associated with tight junction |
| Hepatocyte, MSC, HUVEC | Expression of liver specific proteins and albumin secretion |
| Hepatocyte, MSC, colon cancer HCT116 cell | Albumin secretion, urea formation |
| Hepatoma carcinoma cell | High expression of angiogenic growth factor |
In 2013, Takebe et al. used iPSC-derived hepatocyte-like cells to co-culture with human umbilical vein endothelial cells and human mesenchymal stem cells on Matrigel-coated plates to generate 3D polymers similar to human liver bud tissue, which is the first prototype of iPSC-derived hepatocyte organoids. In 2013, Hans Clevers et al. performed the first study on human liver organoids. They found that by activating the Wnt signaling pathway and using Lgr5+ liver stem cells, liver organoids could be successfully constructed. And through differentiation and transplantation experiments, they proved that these liver organoids have the functional activity of hepatocytes [1]. Although the constructed liver organoids only have some of the basic functions of the liver, they lay the foundation for subsequent research and development.
In 2016, Ma Xuanyi et al. used 3D bioprinting technology to construct iPSC-derived liver organoids [2].
Liver cancer organoids were applied for disease modeling and drug screening in 2017 [3]. The liver is a multicellular organ with complex functions, and single-cell organoids cannot fully recapitulate liver functions. In 2017, Guan et al. used iPSCs to achieve co-differentiation of hepatobiliary cells for the first time, resulting in hepato-biliary organoids. During the same period, Laura Broutier et al. successfully cultured 8 primary liver cancer (PLC) organoids from liver cancer tissue derived from liver cancer patients. The model could not only contain the tissue structure, expression profile, genomic landscape, and in vivo tumorigenicity of the parental tumor but also can be a model for biomarker identification and drug screening. It directly indicated that organoids are useful in identifying genes and potential prognostic values of new therapeutic targets for liver cancer.
In 2018, Hu et al. used primary liver tissue to culture liver organoids with small bile duct structures. In 2018, Sandro Nuciforo et al. successfully cultured liver cancer organoids from puncture samples of patients with advanced liver cancer, and identified the liver cancer organoids after 32 weeks of continuous culture [4]. It was found that the liver cancer organoids still maintained the genomic characteristics and genetic heterogeneity of the primary tumor.
In 2019, Takebe et al. successfully constructed three interconnected organoids of the liver, pancreas and bile duct using iPSC for the first time in the world, which provided the possibility to study the complex interaction process in the early formation of human organs [5]. In 2019, Sun, L. et al. [6] used recombinant human stem cells with inactivated p53 and Rb to culture organoids with liver structure and function. And through genetic engineering technology, they observed that the overexpression of c-Myc can lead to carcinogenesis of liver organoids. By analyzing enriched mutations in human intrahepatic cholangiocarcinoma cells, they demonstrated that combined inhibition of Notch and JAK-STAT prevented RAS-induced lineage transition of hepatocytes to intrahepatic cholangiocarcinoma cells.
In 2020, Guan et al. identified a pathway that is critical for early liver development, phosphatidylethanolamine biosynthesis, through liver organoid modeling [7]. Based on this discovery, they developed a new combination drug for treating primary liver cancer.
References:
[1] Meritxell Huch, Craig Dorrell, Hans Clevers, et al. In vitro expansion of single Lgr5+ liver stem cells induced by Wnt-driven regeneration [J]. Nature. 2013 Feb 14; 494(7436): 247–250.
[2] Ma Xuanyi, Qu Xin, Zhu Wei, et al. Deterministically patterned biomimetic human iPSC-derived hepatic model via rapid 3D bioprinting [J]. Proc Natl Acad Sci, 2016, 113: 2206-2211.
[3] Broutier L, Mastrogiovanni G, Verstegen MM, et al. Human primary liver cancer-derived organoid cultures for disease modeling and drug screening [J]. Nature Medicine, 2017, 23(12): 1424-1435.
[4] Sandro, N., et al. Organoid Models of Human Liver Cancers Derived from Tumor Needle Biopsies [J]. Cell Reports, 2018. 24(5): p. 1363-1376.
[5] Koike H, Iwasawa K, Ouchi R, et al. Modelling human hepatobiliary-pancreatic organogenesis from the foregut-midgut boundary [J]. Nature, 2019, 574(7776) : 112-116.
[6] Sun, L., et al. Modelling liver cancer initiation with organoids derived from directly reprogrammed human hepatocytes [J]. Nature Cell Biology, 2019. 21(8): p. 1015-1026.
[7] Guan Yuan, Chen Xinyu, Wu Manhong, et al. The phosphatidylethanolamine biosynthesis pathway provides a new target for cancer chemotherapy [J]. Hepatology, 2020, 72(4): 746-760.
Most Common Organoids
Related Articles